Investors
BXQ-350 provides a value proposition that is possibly unprecedented
in how it may address significant unmet medical need across these patient populations suffering from devastating malignant disease.
BXQ-350 is a Phase 2a clinical-stage, novel, first-in-class biologic
that has a unique mechanism of action, sphingolipid metabolism modulation which favorably activates multiple enzymes, thereby providing the potential for direct anti-tumor efficacy across a variety of advanced solid tumors and neurological disease.
Metastatic Colorectal Cancer patients continue to have an inferior prognosis despite the use of Standard of Care (SOC) (FOLFOX + bevacizumab). Very few therapies have been approved in first line mCRC in the past decade
In mCRC, mPFS (median Progression Free Survival) averages 9-10 months and mOS (median Overall Survival) is 22-24 months with SOC (FOLFOX + bevacizumab).
FOLFOX is a chemotherapeutic regimen containing Oxaliplatin which is commonly associated with major dose-limiting toxicity (DLT) resulting in reduction in dose, skipping a dose or removal of Oxaliplatin in these patients.
Pipeline in a product
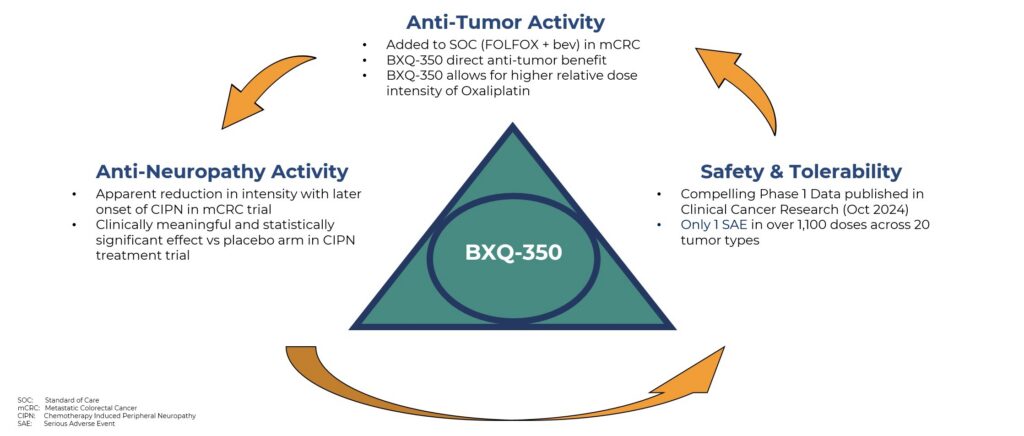
BXQ-350 is the first in a new class of therapies
that has the potential to address unmet medical needs in terms of anti-tumor efficacy but also in terms of anti-neuropathy effect

Non-cancer indications
While BXQ-350 was developed to target solid tumors specifically, early efficacy in neurological disease has emerged.
Addressing an unmet medical need
BXQ-350 has potentially unprecedented anti-tumor and anti-neurological disease effects that show promise in treating cancer patients whose options for new therapies have historically been limited.
Resources
Find case studies, abstracts, and more about the potential breakthroughs we’re pursuing