Bexion Pharmaceuticals, Inc. Announces Publication of Pediatric Phase 1 (KOURAGE) Results
Study achieved planned maximum dose of BXQ-350 with strong safety profile
FOR IMMEDIATE RELEASE Covington, KY, February 23, 2022
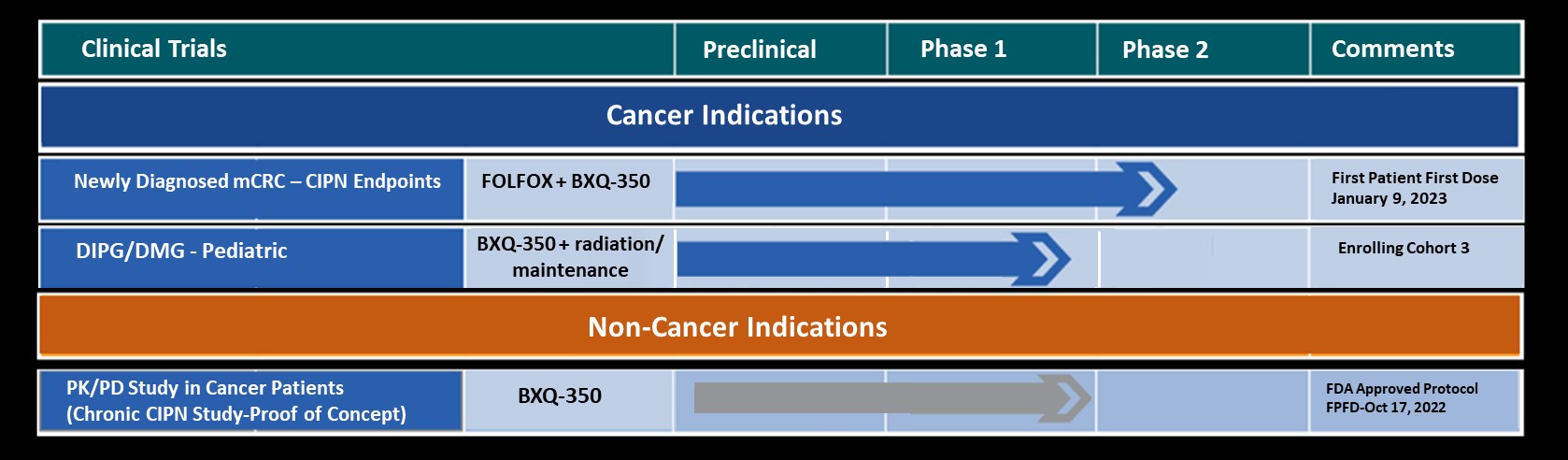
Bexion Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company developing biologics for the treatment of cancer and neuropathy, announced today results from their clinical trial entitled, “An open-label multi-center phase 1 safety study of BXQ-350 in children and young adults with relapsed solid tumors, including recurrent malignant brain tumors (KOURAGE)” have been published in the Volume 8, Issue 12, December, 2022, e12450 issue of Heliyon.
Highlights of this study included:
- A review of the Phase 1 dose escalation study of BXQ-350 in pediatric solid tumors including CNS.
- BXQ-350 is a well-tolerated intravenous drug and achieved the Maximum Planned Dose.
“Results from this pediatric phase 1 study reinforces the excellent safety profile we observed in our adult phase 1 study of BXQ-350 in advanced solid tumors and recurrent high-grade glioma,” stated Scott Shively, President and CEO. “Our safety and maximum planned dose objectives were met in this study, enabling Bexion to open a pediatric phase 1 study in newly diagnosed diffuse intrinsic pontine glioma (DIPG) and diffuse midline glioma (DMG), advancing our commitment to pediatric cancer research.”
About Bexion Pharmaceuticals
Bexion Pharmaceuticals, a clinical-stage biopharmaceutical company, is developing a new generation of biologic immunotherapy to treat solid tumor cancers and Chemotherapy Induced Peripheral Neuropathy (CIPN) with potential portfolio expansion opportunities in other cancers and broader neuropathic pain indications. Bexion’s lead drug candidate is BXQ-350, a first-in-class biologic containing the multifunctional, lysosomal activator protein, Saposin C and a phosphatidylserine.
BXQ-350, an S1P Activator, has demonstrated pre-clinical antitumor effects in vitro and in vivo, particularly in colorectal, brain and other solid tumors. Bexion has completed two single agent Phase 1 programs in adults and in a pediatric population. The Phase 1 programs demonstrated a strong safety profile with evidence of single agent activity across a range of tumors including Glioblastoma Multiforme (GBM), colorectal cancer and appendiceal cancer. Additionally, other clinical and non-clinical data suggest BXQ-350 has activity in chemotherapy induced peripheral neuropathy.
Media Contact: Margaret van Gilse ●859.757.1652 ● [email protected].