Press Releases
Bexion Pharmaceuticals Announces the Addition of Olivia F. Kirtley to Board of Directors
FOR IMMEDIATE RELEASE Covington, KY February 22, 2021 Bexion Pharmaceuticals, Inc. announced today that it has added Olivia F. Kirtley to its Board of Directors. Ms. Kirtley, a Certified Public Accountant and Chartered Global Management Accountant, is a business consultant on strategic, risk and corporate governance issues. She brings to Bexion extensive experience as a former chief financial officer of an international [...]
Bexion Pharmaceuticals Announces Database Lock in Phase 1 Trial of BXQ-350
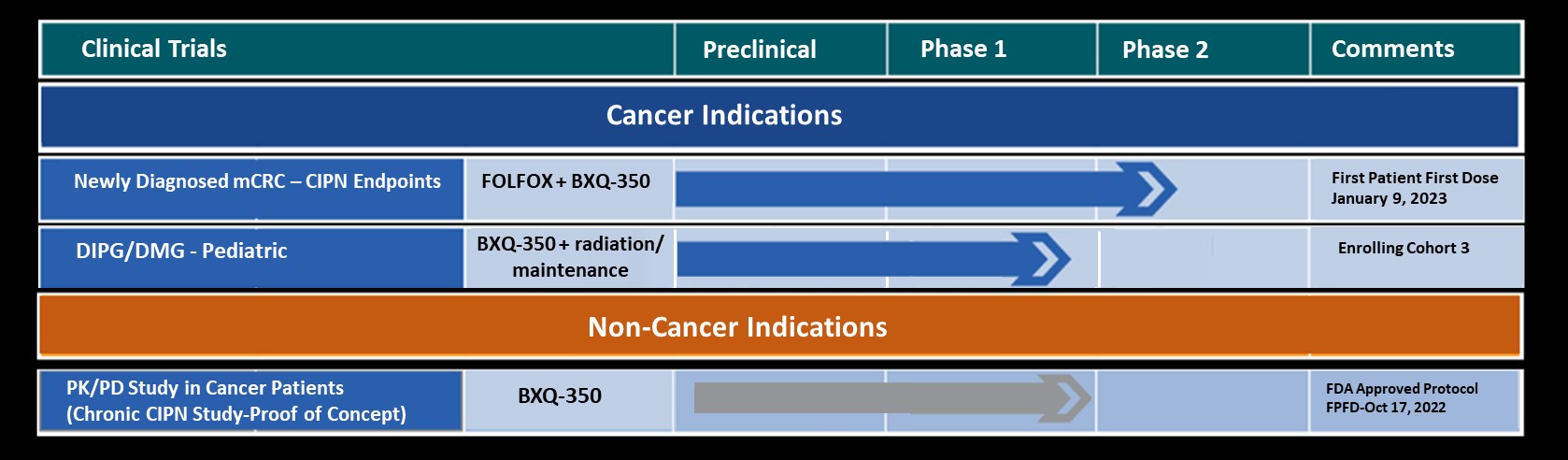
FOR IMMEDIATE RELEASE Covington, KY, December 10, 2020 Bexion Pharmaceuticals, Inc., a clinical stage biotech company focused on the discovery and development of life-changing treatments by leveraging the lysosome's roles in solid tumors and cell death with an initial focus on cancer, announced today the database lock for the Phase 1 study of BXQ-350 in adult patients with advanced solid tumors. Bexion presented interim results [...]
Bexion Pharmaceuticals Announces the Addition of Dr. Raymond J. Tesi to Board of Directors
FOR IMMEDIATE RELEASE Covington, KY November 16, 2020 Bexion Pharmaceuticals, Inc. announced today that it has added RJ. Tesi, M.D. to its Board of Directors. Dr. Tesi has a distinguished career as a biotech entrepreneur and an experienced surgeon. Currently, he is President, Chief Executive Officer and acting Chief Medical Officer of INmune Bio (NASDAQ: INMB). Prior to his role at INMB, he [...]
Bexion Pharmaceuticals, Inc. Receives Rare Pediatric Disease Designation
FOR IMMEDIATE RELEASE [Covington, KY] September 30, 2020 Bexion Pharmaceuticals, Inc., a clinical stage biotechnology company focused on developing innovative cures for cancer, announced that the Federal Drug Administration (FDA) has granted the company’s request for rare pediatric disease designation for BXQ-350, Bexion’s lead therapeutic, for the treatment of diffuse intrinsic pontine glioma (DIPG) and diffuse midline glioma (DMG). Rare Pediatric Disease designation [...]
Bexion Pharmaceuticals, Inc. to Present Clinical and Pre-Clinical Data at 2020 SNO Annual Meeting
FOR IMMEDIATE RELEASE [Covington, KY: September 16, 2020] Bexion Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company focused on the development and commercialization of innovative cures for cancer announced today it will present clinical and pre-clinical electronic posters with audio presentations at the Society of Neuro-Oncology (SNO) annual virtual conference to be held November 19-21, 2020. "We are excited to share these latest clinical and [...]
Bexion Pharmaceuticals Presenting BXQ-350 Data at ASCO 2020
For Immediate Release, Covington, KY May 13, 2020 Bexion Pharmaceuticals announced today that the following three abstracts submitted to the 2020 American Society of Clinical Oncology (ASCO) have been accepted for oral or poster presentation: Oral Presentation Tolerability and preliminary efficacy of BXQ-350 for refractory solid tumors and high-grade gliomas: First-in-human, first-in-class phase I trial. Session: Developmental Therapeutics—Molecularly Targeted Agents and Tumor Biology [...]